Free Online Help: Given the following delta H values H2+1/2O2--->H2O delta H =-285.8 H2O2---->H2+O2 delta H = 187.6 Calculate delta H rxn for the following reaction H2O2--->H2O + 1/2O2

SOLVED: Calculate ΔG° for the reaction H2O(g) + 1/2O2(g) = H2O2(g) at 603 K using the following data: H2(g) + O2(g) = H2O2(g) K = 1.21037 at 603 K 2H2(g) + O2(g) =
I) H2O2 + O3 → H2O + 2O2 (II) H2O2 + Ag2O → 2Ag + H2O + O2 Role of hydrogen peroxide in the - Sarthaks eConnect | Largest Online Education Community
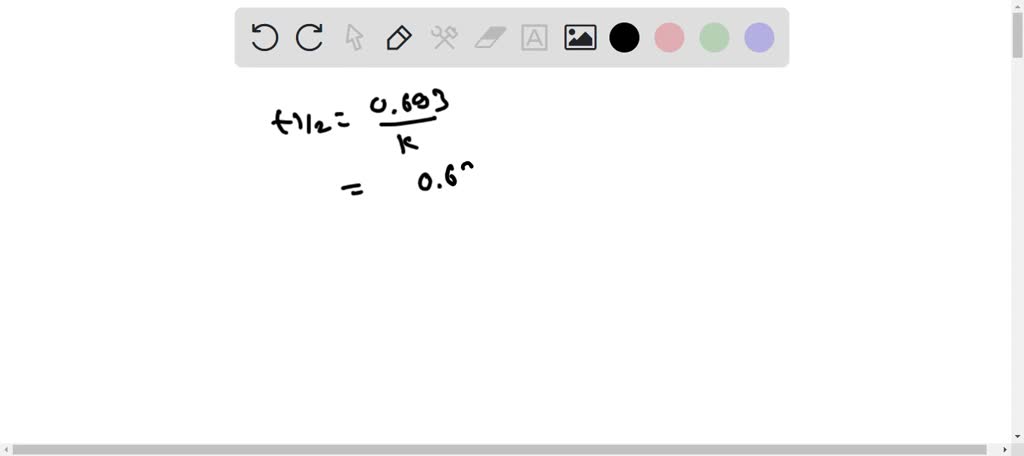
SOLVED: H2O2(aq) ⟶ H2O(l) + 1/2O2(g) a) The above chemical equation is for a first-order reaction. At 303 K, the rate constant equals 9.4 × 10^(-4) s^(-1). Calculate the half-life at this

16.38d | Would hydrogen peroxide be a suitable candidate for fuels: H2O2(l) → H2O(g) + 1/2O2(g) - YouTube

Section B Enthalpy of formation & Enthalpy of combustion and Bomb calorimeter The enthalpy of the reaction H202(0)+ H2O(+ 1/2O2(g) is - 23.5 kcal mol and the enthalpy of fornuation of 120 ()

2H202 alkaline medium *2H20 + 02 the proposed mechanism is as given below : (1) H2O2 +1 → H2O+IO (slow) (2) H202 + 10 + H20+1+02 (fast) (i) Write rate law the

Determine enthalpy of formation H2O2(1), using listed enthalpies of reaction: N2H4(1) + 2H2O2(1) → N2(g) + 4H2O(l); AH; =-818 kJ/mol N2H4(1) + O2(g) → N2(g) + 2H2O(1); 4.H; = -622 kJ/mol H2(g) +


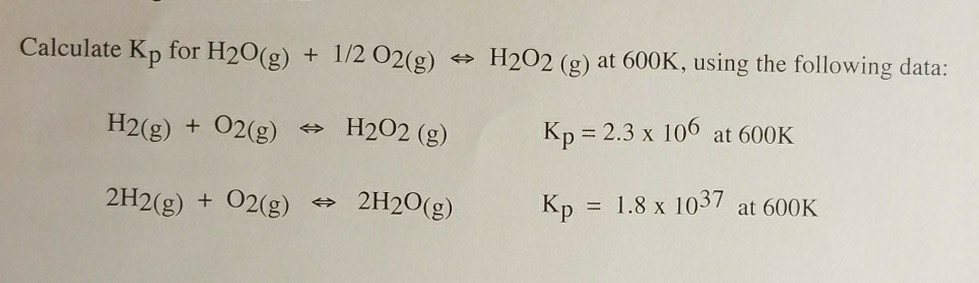



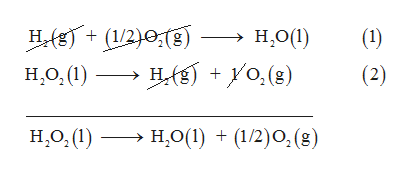

![Odia] H2O2 to H2O + 1/2 O2 is order reaction. Odia] H2O2 to H2O + 1/2 O2 is order reaction.](https://static.doubtnut.com/ss/web-overlay-thumb/11729098.webp)




